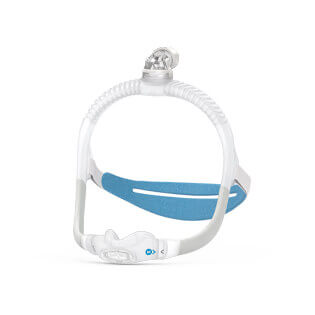
AirFit™ P30i
Nasal pillows mask
Ultra-compact, tube-up mask with a nasal pillows cushion so you can sleep comfortably in any position and get close to your bed partner. The slim, minimalist AirFit P30i features a QuietAir vent, a self-adjusting SpringFit frame and a modern tube-up design.




Sleep close in comfort

The AirFit P30i is designed for closeness. When you want to cuddle up, you can. The QuietAir vent gently diffuses air to limit noise and drafts.1 The top-of-the-head design means there’s no tube to get in the way of a goodnight kiss.
Sleep freely all night long

The clever tube-up design keeps your CPAP tubing out of the way and means it can move fluidly with every twist and turn. The self-adjusting SpringFit frame and dual-wall nasal pillows cushion provide a stable, reliable fit.1, 2 Together, these features offer you the freedom to move easily and sleep in any position you choose, all night long.
Shop online
Need new equipment, accessories or spare parts? It’s quick and convenient to shop on the ResMed online store from anywhere, at any time. Register for our membership programme and you’ll also receive information on our latest products and promotions.
How do I fit my AirFit P30i mask?
How do I assemble/disassemble my AirFit P30i mask?
How do I clean my AirFit P30i mask?
Interested in exploring other mask options?
How do you sleep? What makes you feel more comfortable? Our mask categories help you identify a CPAP mask that matches your sleep needs and preferences.
Please refer to the user guides for relevant information related to any warnings and precautions to be considered before and during use of the products.
References:
- ResMed external 14-day clinical study of 23 ResMed patients, conducted between 30/05/2018 – 22/06/2018. Data on file; ID A4387859.
- ResMed external 7-day clinical study of 24 ResMed and non-ResMed patients, conducted between 21/02/2018 – 12/03/2018. Data on file; ID A4320645.