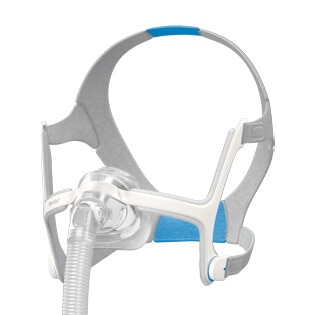
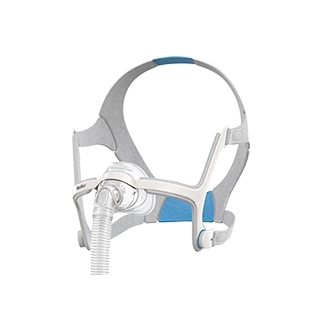
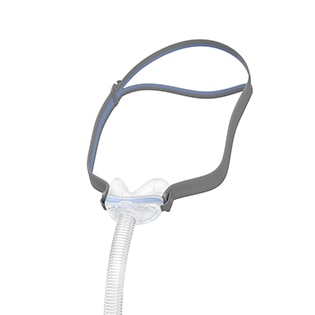
AirTouch™ N20
A soft nasal mask for CPAP therapy
The AirTouch N20 nasal mask features a soft memory foam cushion that is designed to be comfortable and limit red marks around the nose.1 Compatible with the AirFit N20 CPAP mask,2 you can use both cushions in the same mask frame
The mask contains magnets that may interfere with certain implants or medical devices. Please refer to the user guide for complete information, including magnet contraindications and warning.

A comfortable memory foam cushion
Designed to limit red marks1 around the nose, the AirTouch N20 memory foam cushion can adapt to your individual facial features. If you are looking for a full face mask featuring the memory foam cushion, visit the AirTouch F20 full mask product page.

Mix and match with the AirFit N20
The AirTouch N20 memory foam cushion and the AirFit N20 silicone cushion are interchangeable, allowing you to swap between the two as you like.2 Each size of cushion easily clicks into the same mask frame.
Shop online
Need new equipment, accessories or spare parts? It’s quick and convenient to shop on the Resmed online store from anywhere, at any time. Register for our membership programme and you’ll also receive information on our latest products and promotions.
Please refer to the user guides for relevant information related to any warnings and precautions to be considered before and during use of the products.
Contraindication
Masks with magnetic components are contraindicated for use by patients where they, or anyone in close physical contact while using the mask, have the following:
- Active medical implants that interact with magnets (i.e., pacemakers, implantable cardioverter defibrillators (ICD), neurostimulators, cerebrospinal fluid (CSF) shunts, insulin/infusion pumps)
- Metallic implants/objects containing ferromagnetic material (i.e., aneurysm clips/flow disruption devices, embolic coils, stents, valves, electrodes, implants to restore hearing or balance with implanted magnets, ocular implants, metallic splinters in the eye)
Warning
Keep the mask magnets at a safe distance of at least 6 inches (150 mm) away from implants or medical devices that may be adversely affected by magnetic interference. This warning applies to you or anyone in close physical contact with your mask. The magnets are in the frame and lower headgear clips, with a magnetic field strength of up to 400mT. When worn, they connect to secure the mask but may inadvertently detach while asleep. Implants/medical devices, including those listed within contraindications, may be adversely affected if they change function under external magnetic fields or contain ferromagnetic materials that attract/repel to magnetic fields (some metallic implants, e.g., contact lenses with metal, dental implants, metallic cranial plates, screws, burr hole covers, and bone substitute devices). Consult your physician and manufacturer of your implant / other medical device for information on the potential adverse effects of magnetic fields.
References:
- Resmed external clinical study of 26 patients for 7 nights, conducted between 15/03/2018 and 05/04/2018. Data on file; ID A4575326.
- AirTouch N20 user guide, Resmed Pty Ltd 2019. ID A4670547.
Content last updated: 10/2023.