The EPiC-HFT trial
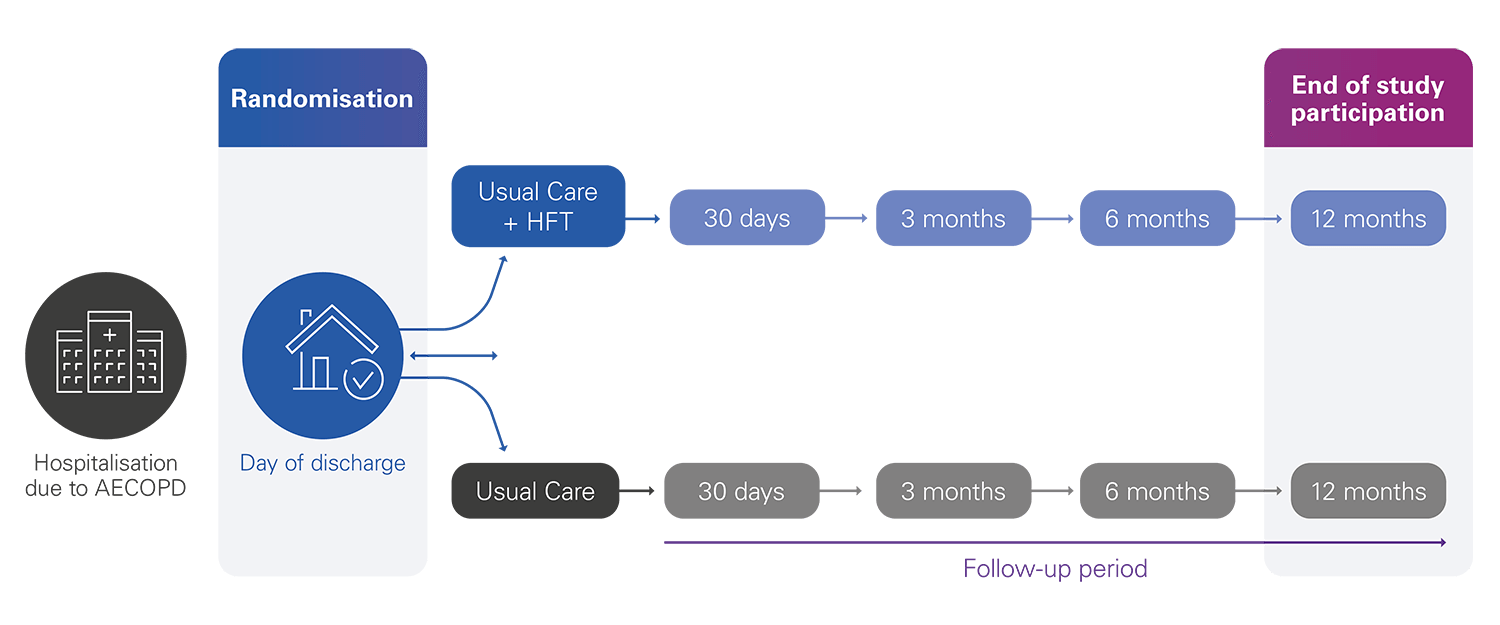
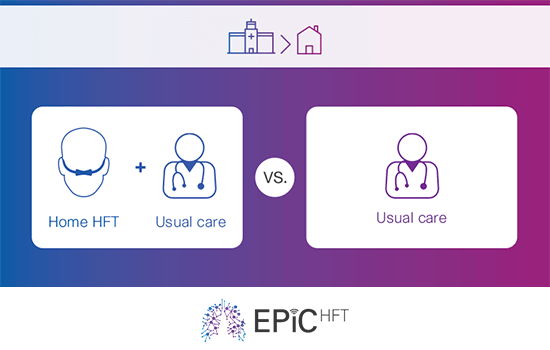
This clinical trial aims to demonstrate the clinical and cost-utility of home high-flow therapy (HFT), assessing the viability of home HFT as a suitable therapy for patients who have previously been hospitalised due to severe chronic obstructive pulmonary disease (COPD) exacerbation.
Study objectives*
The main objective of the trial is to investigate whether home HFT improves 12-month admission-free survival times, following a severe exacerbation of COPD requiring hospitalisation.

Patient population
- Patients admitted to hospital with an exacerbation of COPD
- Age 40-80 years with smoking history >10 pack years
- Patient’s forced expiratory volume in 1 second (FEV1) is <80% predicted
- Patient’s FEV1/forced vital capacity (FVC) < 0.7
The study endpoints
Primary endpoint:
12-month all cause admission-free survival
Secondary endpoints:
- Patient reported outcome measures (CAT, PSQI, EQ-5D-5L)**
- Physiological effects of HFT
- Cost utility analysis
Join the Resmed Community
Stay connected and you’ll be among the first to hear all the latest news and developments.
This content is intended for health professionals only
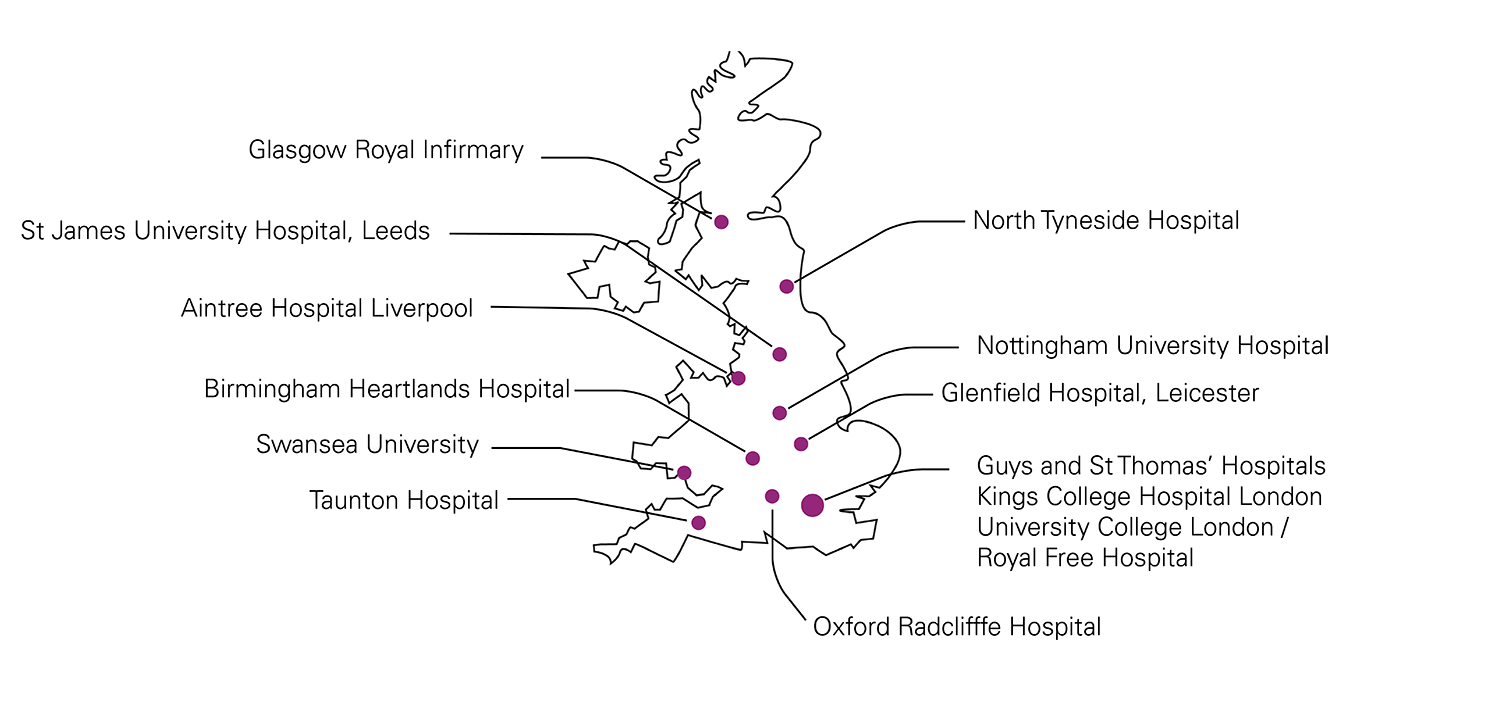
*For more information visit the investigator’s research page https://www.kcl.ac.uk/research/epic-hft
**AECOPD, acute exacerbation of chronic obstructive pulmonary disease; BMI, body mass index; CAT, computerised adaptive tests; COPD, chronic obstructive pulmonary disease; EPIC, exacerbation prevention in COPD using home high flow therapy; EQ-5D-5L, EuroQol 5-dimension, 5-level quality of life questionnaire; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; HFT, high flow therapy; NIV, non-invasive ventilation; OSA, obstructive sleep apnoea; PaCO2, partial pressure of carbon dioxide; PAP, positive airway pressure; PSQI, Pittsburgh sleep quality index.
Content last updated: 03/2024